Forensic Intelligence for Clinical Trial Capital
We surface what stalled trials, silent sponsors, and "failed" assets are actually telling you — before your next $1M/day decision

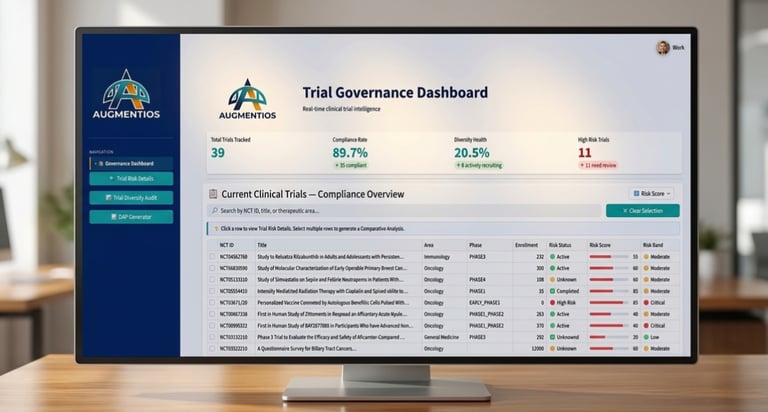
The $1M/Day Decision Problem
Every day a stalled trial sits in your pipeline — or a competitor's silent asset sits unchallenged — capital burns. Augmentios shortens the gap between signal and action.
The DAPORT Engine
A purpose-built forensic intelligence platform that reads what clinical trial registries don't tell you. Reporting Gaps. Operational decay. Mechanistic fingerprints. Regulatory exposure. All scored, all sourced, all actionable.
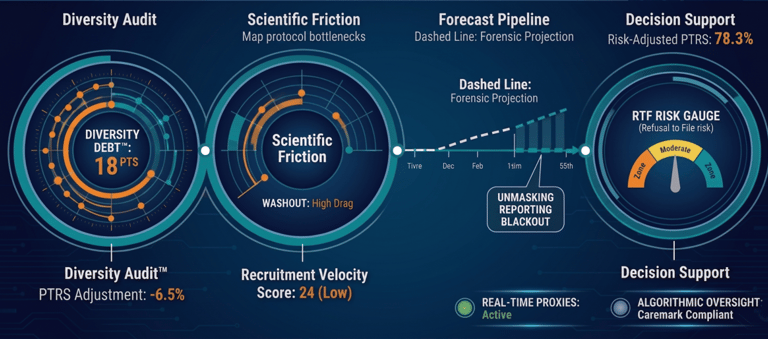
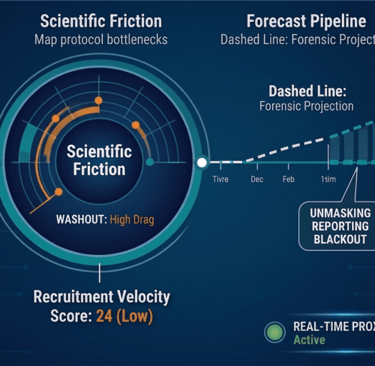


The Forensic Suite
A unified intelligence blueprint designed for high-stakes M&A, competitive strategy, and clinical capital protection.
Strategic Command Center
Real-time surveillance of global trial portfolios, reporting-gap detection, and automated execution scoring.
Forensic Asset Auditing
Deep-learning audits that unmask the "Truth Gap" in individual trials to verify mathematical defensibility for M&A.
Automated assembly of mission-critical FDA requirements to eliminate FDORA holds and secure first-to-market status.
Regulatory Defense Engine

Competitive Intelligence That Sees Around Corners
Most competitive intelligence tools tell you a competitor's trial exists. They don't tell you it stopped reporting 147 days ago, that enrollment velocity decayed 60% before the silence began, or that the sponsor's last three press releases stopped mentioning the program by name.
DAPORT does.
For competitive intelligence, forecasting, and brand strategy teams who need to know — before the conference call — which competitor programs are real, which are stalling, and which are quietly being shelved.
✓ Reporting Gap surveillance across every competitor program in your therapeutic area, refreshed continuously
✓ Operational decay scoring that quantifies how badly a silent trial has likely slipped
✓ Strategic Leverage Windows — the time-bound openings where a competitor's stall becomes your accelerated launch
✓ Indication-expansion scouting — find the trials your competitors aren't running in populations that matter
✓ Investor-grade evidence — every signal sourced, timestamped, and defensible to your CFO and your board
Users of DAPORT
Biotech and Pharma
Your competitors' silent trials are your loudest market signal. DAPORT continuously scans the global registry, flags Reporting Gaps, scores operational decay, and surfaces Strategic Leverage Windows — so your BD, clinical, and corp dev teams move on the same intelligence.
Used for: competitive trial monitoring · indication expansion scouting · DAP generation for your own programs · investor-grade risk briefs
CROs and Regulatory
Hospitals and Academic Centers
Investors and Capital Allocators
Win more sponsor mandates with forensic intelligence baked into your pitch. DAPORT lets you walk into a kickoff already knowing the sponsor's Reporting Gap exposure, FDORA diversity gaps, and competitive landscape — then generate the defensible DAP in the same workflow.
Used for: sponsor pitches · feasibility studies · diversity strategy · post-award compliance delivery
See which trials in your therapeutic franchise are stalling, which sponsors are silent, and which underserved patient populations represent your strongest enrollment leverage. DAPORT helps academic medical centers and integrated delivery networks identify where their patient catchment is the strategic answer to a sponsor's gap.
Used for: site selection bids · franchise gap analysis · community trial design · sponsor outreach evidence
Before you commit to a Series B, an asset acquisition, or a licensing deal — see what the trial registry is whispering. DAPORT delivers forensic dossiers on target programs: Reporting Gap history, operational decay scoring, regulatory exposure, comparable-trial benchmarking, and diversity-compliance posture. The questions your IC should be asking, answered before the management meeting.
Used for: pre-IC diligence · asset acquisition screens · portfolio monitoring · Rescue & Repurposing target identification








We will send you a 1-page Forensic Read at no cost

The Augmentios Difference: Precision Arbitrage
The $1M/Day Race to Market
In the current "Lower Ceiling" environment, market leadership is determined by Execution Velocity. A single day of delay is a $1M NPV loss. While legacy competitors sell reactive "rescue" services and open-ended consulting, Augmentios provides Forensic Clinical Intelligence. We don't just find the data; we unmask the Reporting Gaps that allow you to leapfrog stalled competitors and capture the first-to-market premium.
Competitive Arbitrage: Turning FDORA into a Moat
Global competition—specifically the surge in low-cost, high-volume drug starts from China—is threatening U.S. biotech returns. But speed without compliance is a trap. Augmentios utilizes Diversity Arbitrage to protect your assets. While competitors accumulate "Diversity Debt" that leads to binary FDA Refusal-to-File (RTF) risks, our engine ensures your asset is the most Regulatory-Robust on the market. We use the FDA’s strict mandates as a strategic barrier to entry against non-diverse global competition.
Liquidity through Forensic Certainty
We replace months of "Clinical Archaeology" with 60 minutes of Transaction-Ready Forensics. Whether you are auditing a Chinese-originated asset for the U.S. market or identifying the "Rescue Rationale" in a stalled Phase II, Augmentios provides the mathematical proof required to close the deal. Backed by 30 years of blockbuster clinical leadership, we don't just monitor risk—we mathematically eliminate the obstacles to your next exit.